2-Octyl Cyanoacrylate (Dermabond) Wound Adhesives: Product, Design Features, Indications
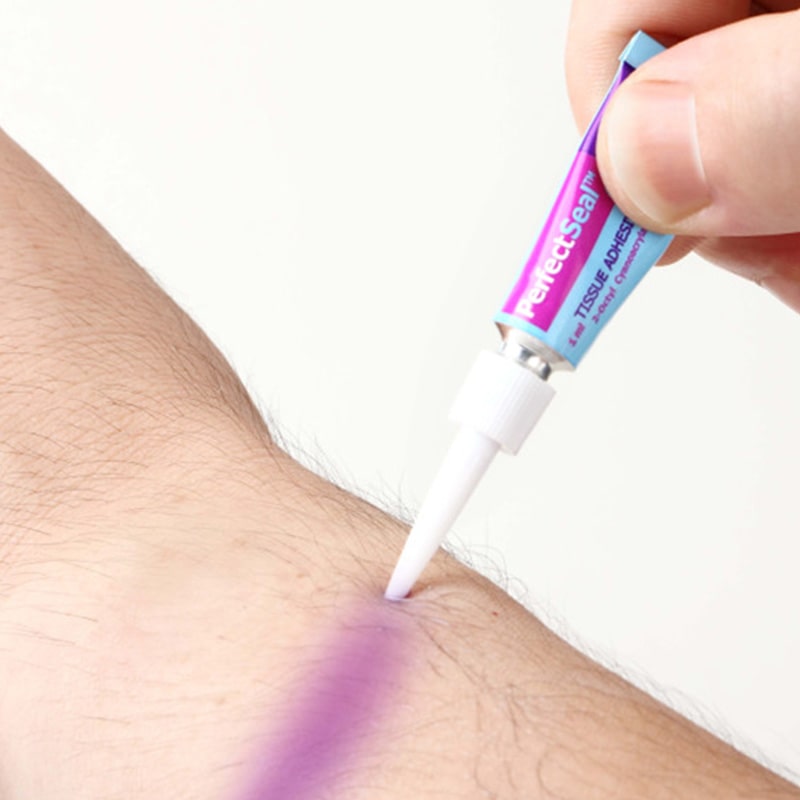
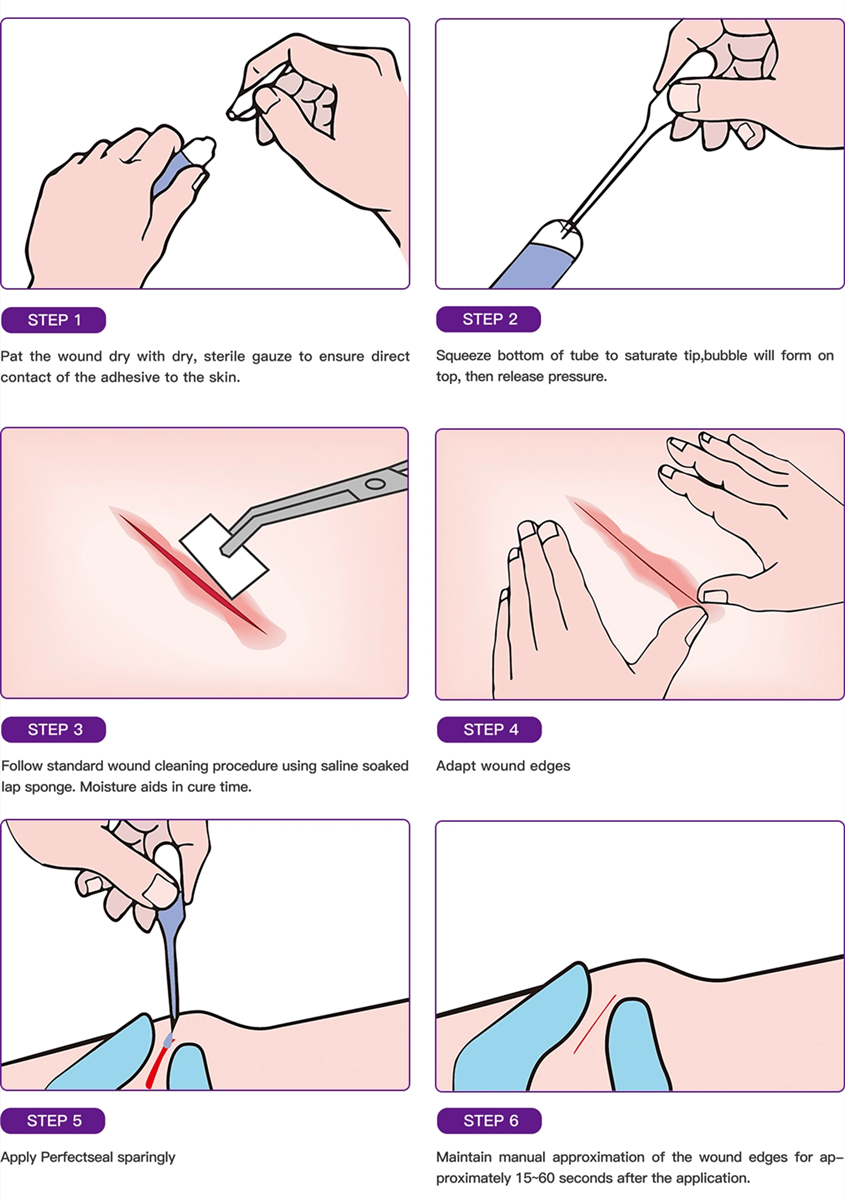
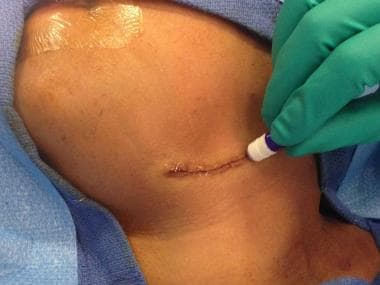
The wound adhesive 2-octyl cyanoacrylate (Dermabond) is approved by the US Food and Drug Administration (FDA) for closure of incised skin. In addition to its surgical adhesive indication, 2-octyl cyanoacrylate was approved by the FDA in January 2001 for use as a barrier against common bacterial microbes, including certain staphylococci, pseud
Ethicon Eth CLR602US Dermabond Prineo Skin Closure System
Cureus A Comparative Study Between Conventional Sutures, Staples

DRSC, Healthcare Professionals
Dermabond Advanced DNX6 Topical Skin Adhesive - Henry Schein Medical

CE ISO Liquid Stitches Octyl Cyanoacrylate CAS#184765-00-2

Allergic Contact Dermatitis to Dermabond Prineo After Elective

PDF) Comparison of 2-octyl cyanoacrylate and n-octyl cyanoacrylate

Topical Skin Adhesive - SourceMark

2-Octyl Cyanoacrylate - an overview